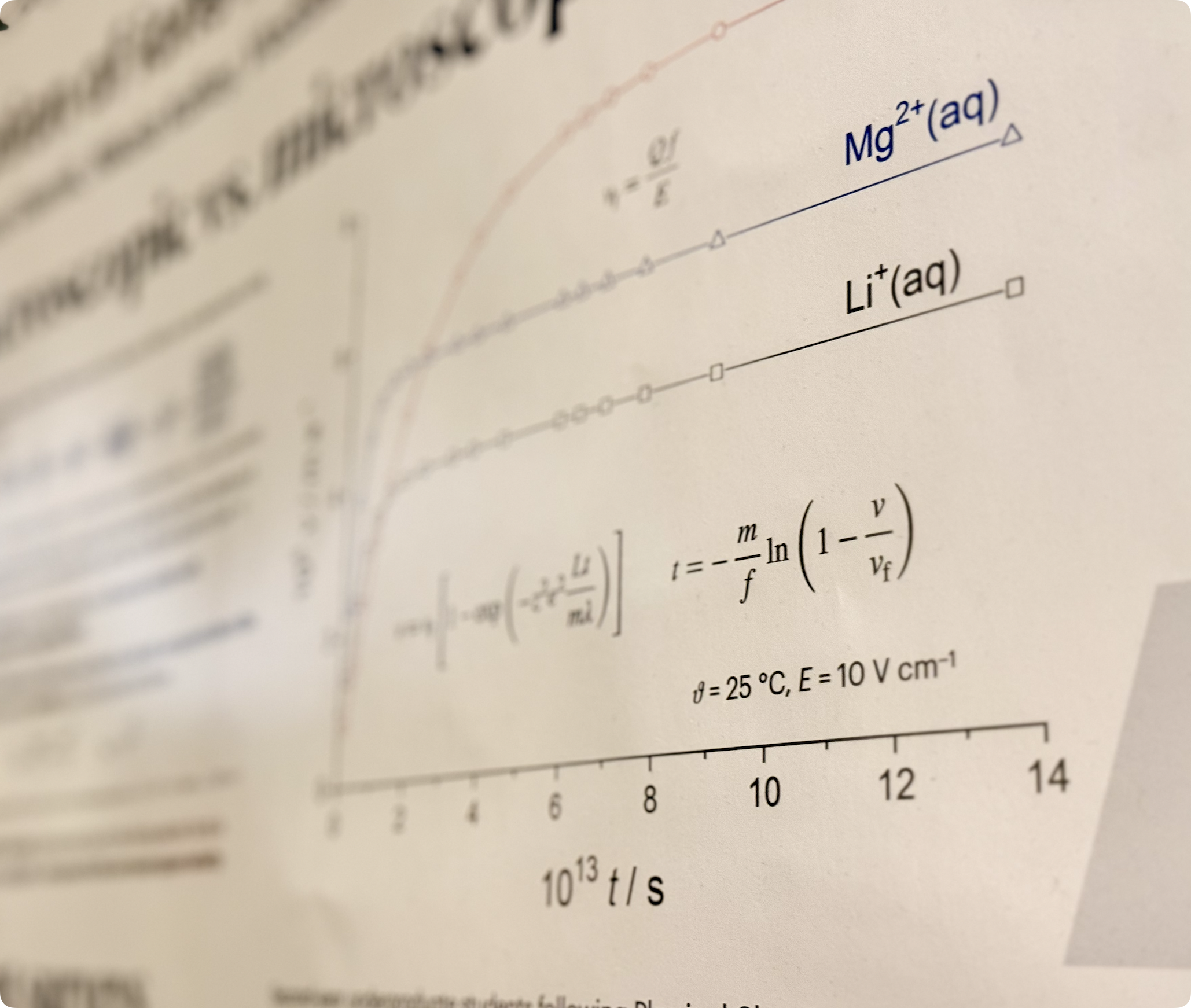Department of Chemistry
The history and development of the Department of Chemistry
The history of the Department of Chemistry begins in 1874 with the founding of the Division of Chemistry at the Faculty of Philosophy, University of Zagreb. In 1876, Aleksandar Velkov gave his first lectures in chemistry. For the purposes of teaching, he opened laboratories located in a small building at 1 Nova Ves, Zagreb. After his untimely death, chemistry was temporarily taught by botanist Bohuslav Jiruš, until Czech scholar Gustav Janeček was appointed as associate professor in 1879. Due to the increased number of students and expansion of the curriculum, Janeček initiated the construction of a new division building at today's 14 Strossmayer Square, which opened in 1884. With such improved conditions, great progress was made in the work of the Division of Chemistry; laboratory exercises were broadened and scientific research was initiated in which more advanced students took part. Particular progress was made in the fields of physical and analytical chemistry. Gustav Janeček strived for decades, as the sole representative of the chemical profession, to extend chemical training, establish departments for individual branches of chemistry, especially analytical and physical chemistry, and divide the department of general and experimental chemistry into the departments of inorganic and organic chemistry. At Janeček’s initiative, the construction of a new building of the Division of Chemistry at 20 Marulić Square began. The building was finished in 1919, but it was not handed over to the Department of Mathematics and Natural Sciences of the Faculty of Philosophy, as originally intended, but was given to the newly established Higher Technical College. After Janeček's retirement in 1921, the Russian chemist Nikola A. Pushin came to the Division and became the Head of the newly-established Subdepartment of Physical Chemistry. After Pushin left Croatia in 1928, Gilbert Flumiani took over the Division, the head of which he remained until the end of World War II. At that time, two departments had already been formed, the Pharmaceutical and the Chemical. During the war and after it ended, many university institutions were reorganized, so the Department of Pharmacy was singled out from the Division of Chemistry in 1942 as the new Faculty of Pharmacy, and in 1946 the Department of Mathematics and Natural Sciences was separated from the Faculty of Philosophy. The Department of Mathematics and Natural Sciences changed name and the newly-established Faculty of Science was organized into six departments. One of them was the Department of Chemistry, which initially consisted of the newly-founded Institute of Chemistry, headed by Krešimir Balenović, and the Physicochemical Institute with Božo Težak as head. The Institute of Chemistry covered all fields of chemistry except physical chemistry: general and inorganic chemistry was taught by Stanko Miholić, organic chemistry and biochemistry was taught by Krešimir Balenović, and analytical chemistry was taught by Ladislav Filipović. Following the first division into the two aforementioned institutes, the other newly-established divisions within the Department separated. Thus, in 1952, the Laboratory of General and Inorganic Chemistry (later changing its name to the Division of General and Inorganic Chemistry) become a new institution, with the new head being Drago Grdenić, and in 1962 the Division of Analytical Chemistry with Ladislav Filipović as head. Already in 1959, the Physicochemical Institute changed its name to the Division of Physics and Chemistry and was headed by Božo Težak. After the separation of individual divisions from the Institute of Chemistry, in 1961 it changed its name to the Division of Organic Chemistry and Biochemistry, headed by Krešimir Balenović. All four divisions began to develop teaching and especially scientific activities.

 Pristupačnost
Pristupačnost